Resistant Dextrin Due Diligence: A Fast Risk-Check Pack for Procurement & Legal
Non‑GMO resistant dextrin (often labeled as soluble corn fiber) has become a cornerstone ingredient in the global functional food, beverage, and dietary supplement markets. Its versatility, neutral taste, and fiber enrichment capabilities make it indispensable. However, for procurement managers and legal reviewers, sourcing this ingredient involves more than just finding a supplier with a competitive price.
The real challenge lies in preventing compliance gaps, quality drift, and Intellectual Property (IP) exposure once the ingredient is integrated into your product formulations, labeling claims, and audit trails. In an era of heightened regulatory scrutiny and complex global supply chains, a robust due diligence process is your best defense against recall risks and legal entanglements.

At Shine Health, we understand these pressures. As a dedicated manufacturer and supplier, we produce premium resistant dextrin from non‑GMO corn starch within GMP-standard workshops. Our process is backed by full batch traceability and a dedicated QC laboratory equipped with advanced testing capabilities. To assist busy procurement and legal teams, we have compiled this essential review pack: a guide on what to verify in a supplier file, which documents to prioritize, and how to mitigate freedom‑to‑operate (FTO) and regulatory risks before signing a contract.
What We Supply: Reducing Sourcing Risk Through Quality
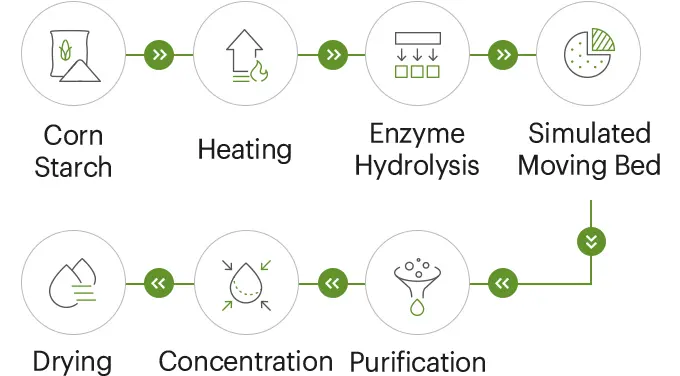
Our resistant dextrin is meticulously produced using a controlled enzymatic hydrolysis process followed by spray‑drying. This method ensures a high-purity, soluble dietary fiber that meets the rigorous demands of modern food processing. For procurement teams, the key value proposition is consistency: a stable specification, a repeatable manufacturing process, and auditable Quality Control (QC) data make the ingredient significantly easier to qualify across diverse product lines—from clear beverages to bakery items.
We offer various grades to suit specific formulation needs, ensuring that you only pay for the specifications you require. Whether you need a standard fiber content or a high-purity isolate, our portfolio is designed to be the reliable backbone of your supply chain.
Technical Baselines and Specifications
When reviewing our resistant dextrin COA (Certificate of Analysis), you will typically see the following strictly controlled parameters. These specs are crucial for ensuring your final product's texture, stability, and nutritional claims:
- Appearance: White to light yellow powder, ensuring no color interference in clear liquids.
- Flavor: Sweet, mild, and pure, allowing for flavor neutrality in sensitive applications.
- Dietary Fiber Content: We adhere to strict grading standards:
- Grade D1: ≥70% Fiber Content
- Grade D2: ≥85% Fiber Content
- Grade D3: ≥90% Fiber Content
- Grade D4: ≥95% Fiber Content
- Moisture: ≤5.0 g/100 g (Critical for shelf stability and flowability)
- Ash: ≤0.1 g/100 g
- pH (10% solution): 3–6 (Compatible with acidic beverage formulations)
- Water Activity: ≤0.2 (Ensures microbial stability)
- Microbiology Limits:
- Aerobic plate count: ≤1000 CFU/g
- Coliforms: ≤3 MPN/g
- Mould & Yeast: ≤25 CFU/g each
To further support your qualification process, our facilities and products carry major international certifications, including ISO9001, BRC, HALAL, HACCP, and KOSHER. These certifications are not just badges; they are active components of our quality management system.
Traceability: Defining "Non‑GMO" in Your Files
In the current market, a generic "non-GMO" claim is often insufficient for rigorous audits. Auditors and consumers alike demand a clean, unbreakable chain of custody from the field to the finished product. For a robust non‑GMO positioning, you must ensure the link between the non‑GMO corn starch input and the finished resistant dextrin lot is transparent.
Your internal onboarding checklist should explicitly confirm:
- Specific Non-GMO Statement: A declaration tied directly to the specific resistant dextrin grade you are purchasing, not a blanket company statement.
- Batch Traceability: The ability to trace a finished goods lot number back to the specific raw material (corn starch) lots used in its production.
- Change-Control Protocols: A clear agreement on notification expectations if the supplier changes enzyme sources, processing parameters, or packaging materials.
Internal Tip: If a supplier’s non‑GMO statement appears generic, request a version that explicitly names "resistant dextrin" or "soluble corn fiber" and references the specific manufacturing site. At Shine Health, our non-GMO corn starch is sourced from premium producers in China, ensuring a reliable and clean starting material.
Reading a Resistant Dextrin COA: The Fast Checklist
Procurement and legal teams do not need to run the wet chemistry methods themselves, but a consistent and intelligent COA review process prevents late-stage surprises that can derail a product launch. Here is how to efficiently scan a COA for red flags.
COA Items That Matter Most
- Dietary Fiber Content: This is the primary value driver. Ensure the testing method (e.g., AOAC 2001.03) aligns with your labeling requirements to prevent "label math" errors.
- Moisture & Water Activity: High moisture can lead to clumping in powder blends and microbial risks. Our strict limit of ≤5.0% moisture ensures excellent stability.
- Microbiological Limits: Verify that the supplier's limits align with your internal specifications. Our strict controls on yeast, mold, and coliforms are designed for sensitive applications like dairy and infant nutrition.
- Appearance & Odor: Any deviation here can be an early warning sign of processing inconsistencies or improper storage conditions.
When to Add Third‑Party Verification
While we maintain a fully equipped internal QC laboratory, we encourage transparency. You should consider independent third-party testing when you:
- First qualify a new resistant dextrin supplier.
- Switch grades (e.g., moving from D2 to D4 for higher fiber claims).
- Enter markets with stricter regulatory frameworks or sensitive consumer populations.
- Use high inclusion levels per serving, where minor impurities could compound.

Market Compliance Map: Essential Documents
Regulatory status and labeling requirements for resistant dextrin vary significantly by region. A "global" launch often requires market-specific documentation. The table below summarizes the most common document requests we fulfill to speed up your approval process.
| Market | What Reviewers Confirm | Documents That Speed Up Approval | Common Risk Flag |
|---|---|---|---|
| United States | Use as dietary fiber; FDA GRAS status correspondence. | Spec + COA, manufacturing description, GRAS letters. | A GRAS letter is often supplier/process specific. Do not assume a competitor's GRAS covers your source. |
| European Union | Fiber use is common; strict control on claim wording. | Label review, supporting dossiers for claims. | Only authorized claims should be used. Avoid unapproved disease prevention wording. |
| China | Use depends on specific food/supplement category rules. | GB compliance statement, label draft, Spec + COA. | Category mismatch can trigger costly label revisions or filing rejections. |
| Japan | Claims require a dossier pathway (e.g., FFC). | Spec + COA, supporting stability/efficacy data. | Claims are not "portable"—brand owners need compliant substantiation for their specific product. |
| India | Fiber claims are strictly controlled by FSSAI. | Spec + COA, compliance statement, label examples. | Claim language must match local tables and definitions exactly. |
| Brazil | Import and labeling checks can be slow. | Portuguese label proof, complete import documentation. | Customs delays often trace back to incomplete or mismatched documentation. |
IP & Freedom‑to‑Operate (FTO): 6 Steps Before Contracting
One of the most overlooked aspects of ingredient procurement is Intellectual Property (IP). Patents around resistant dextrin can cover specific enzyme routes, processing conditions, proprietary steps, or even specific end-use applications. To reduce FTO risk, we recommend a workflow that your legal team can execute in parallel with procurement without slowing down the timeline.

- Patent Landscape Search: Search USPTO, EPO, WIPO, and Google Patents for keywords like "resistant dextrin," "resistant maltodextrin," and "soluble corn fiber."
- Process Mapping: Map claim language in patents against the specific manufacturing process used by your supplier (enzymatic vs. acid hydrolysis, etc.).
- Jurisdiction Check: Flag patents that are still in force in your specific target sales markets.
- Supplier Disclosures: Request explicit disclosures from your supplier regarding owned/licensed patents and any freedom-to-operate analysis they have conducted.
- Contractual Indemnity: Contractually define warranties and IP indemnity scope by market and use case.
- Counsel Opinion: For high‑stakes launches or flagship products, obtain a written opinion from IP counsel.
Why Shine Health? Our Manufacturing Strength
Choosing Shine Health means partnering with a manufacturer that prioritizes technical excellence and supply security. We go beyond simple supply; we offer a partnership built on robust infrastructure:
- Advanced Technology: We utilize a precision production line of German origin, ensuring tight control over particle size and solubility.
- Premium Enzymes: We use advanced biological enzymes imported from overseas to ensure the highest quality conversion and fiber yield.
- Japanese Craftsmanship: Our production philosophy incorporates exquisite craftsmanship techniques from Japan, focusing on detail and purity.
- R&D and ODM: Our robust research and development capabilities allow us to offer ODM services to address diverse requirements, from custom density powders to specific solution clarities.
- 24/7 Support: Our engineers offer round-the-clock online support to resolve technical issues, such as tablet coating challenges or formulation stability questions.
The Minimum "Audit-Ready" Document Pack
If you need to qualify a resistant dextrin supplier quickly, treat the following list as your practical baseline. A supplier who cannot provide these promptly is a risk factor.
- Current Specification Sheet + most recent COA (matching the grade you intend to buy).
- Manufacturing Process Description (including the specific non‑GMO statement).
- GMP/HACCP Evidence and valid certificates (ISO9001, BRC, HALAL, HACCP, KOSHER).
- Regulatory Correspondence applicable to your launch plan (e.g., GRAS self-affirmation summaries where available).
- Patent/Certification Materials relevant to your internal IP review.
For samples, detailed technical data, or a complete document set aligned to your onboarding workflow, please contact us directly. We are ready to support your due diligence process with total transparency.
FAQ
Q: What is the primary raw material for Shine Health's resistant dextrin?
A: Our resistant dextrin is crafted from premium non-GMO corn starch sourced from trusted producers in China. We ensure strict traceability back to the source.
Q: Can you provide different grades of resistant dextrin based on fiber content?
A: Yes, we supply multiple grades to meet different regulatory and formulation needs: D1 (≥70%), D2 (≥85%), D3 (≥90%), and D4 (≥95%).
Q: Is your resistant dextrin suitable for vegetarians and vegans?
A: Yes, our product is plant-based, derived solely from corn starch, and is suitable for vegetarian and vegan diets. It is also Halal and Kosher certified.
Q: How does your product perform in beverages?
A: It exhibits excellent solubility and stability. It dissolves clear in water, has a neutral taste, and remains stable across a wide pH range (3–6), making it ideal for functional beverages.
Q: Do you offer technical support for formulation challenges?
A: Absolutely. We provide 24/7 engineering support to assist with any technical issues, including application advice for tablets, gummies, dairy, and bakery products.
Q: How can I request a quote or sample?
A: You can contact us via email at info@sdshinehealth.com or reach out on WhatsApp at +86 13405443339.
References
- Mysonhimer, A. R., & Holscher, H. D. (2022). Gastrointestinal effects and tolerance of nondigestible carbohydrate consumption. Advances in Nutrition. https://doi.org/10.1093/advances/nmac094
- European Food Safety Authority. (2015). Scientific opinion on a resistant dextrin-type fiber and post-prandial glycaemic response. EFSA Journal, 13(1), 3839. https://www.efsa.europa.eu/en/efsajournal/pub/3839
- U.S. Food and Drug Administration. (n.d.). Agency response letters for GRAS notices (inventory). https://www.fda.gov/food/gras-notice-inventory/agency-response-letter-gras-notice
- Hasjim, J., Ai, Y., & Shi, Y.-C. (2013). Novel applications of amylose–lipid complex as resistant starch type 5. In Starch (chapter reference). https://doi.org/10.1002/9781118528723.CH04
- Matsumoto, M., Fujita, A., et al. (2016). Food and drink product containing poorly digestible compound and colonic-hydrogen-gas producing agent. Patent literature. https://www.semanticscholar.org/search?q=Food%20and%20drink%20product%20containing%20poorly%20digestible%20compound%20and%20colonic-hydrogen-gas%20producing%20agent&sort=relevance
- FoodNavigator-USA. (2023). Multifunctional FiberSMART granted GRAS status (industry report). https://www.foodnavigator-usa.com/Article/2023/04/21/multifunctional-fibersmart-granted-gras-status/
- Food Dive. (2023). FiberSMART sweetener granted GRAS (industry report). https://www.fooddive.com/news/fibersmart-sweetener-gras-tapioca-starch/645750/


